Molar mass 146.14 g/mol Density 1.37 g/cm³ | Formula C9H6O2 Appearance Yellow solid | |
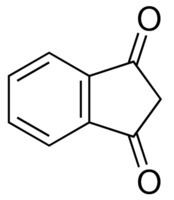 | ||
1,3-Indandione (indanedione) is an organic compound with the molecular formula C9H6O2. Chemically, it is a β-diketone. In standard conditions it is referred to in different sources as either a colorless or yellowish, green, or (most commonly) yellow solid.
Contents
Structural properties
In the solid state, 1,3-indandione occurs as a diketone; in water, it is partially (~2%) enolized. The enolate anion exhibits significant delocalization, and the highest electron density is on the second carbon. This explains many of chemical properties of the compound.
Preparation
1,3-Indandione can be prepared by decarboxylation of the sodium salt of 2-etoxycarbonyl-1,3-indandione, which itself is obtained by Claisen condensation of ethyl acetate and dimethyl phthalate.
Chemical properties
1,3-Indandione is a very strong C-nucleophile. It undergoes self-condensation quite easily, resulting in bindone.
Bromination occurs at the 2-position:
1,3-Indandione could be reduced to indanone, 3-hydroxy-1-indanone, 1,3-indanediol or even indane, depending on the method used.
Related compounds
The related compound 1,2-indandione is used in the first stage of forensic identification of latent fingerprints. It is particularly useful for paper, and for items printed with thermal inks such as receipts. Amino acids left behind by the human hand may be developed into fingerprints by the use of it; the results, photographed with a special filter under a strong yellow-green fluorescent or green laser. It is usually the first method employed in a sequential analysis aimed at the production of evidence of a grade suitable for use in the courtroom. Nivimedone is an antiallergen.
