Molar mass 114.093 g/mol Density 1.16 g/cm³ Appearance colorless liquid | Formula C6H4F2 | |
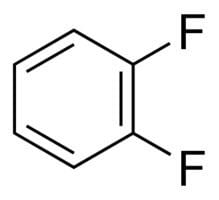 | ||
1,2-Difluorobenzene, also known as DFB, is an aromatic compound with formula C6H4F2. This colorless liquid is a solvent used in the electrochemical studies of transition metal complexes.
Contents
Synthesis
1,2-Difluorobenzene is prepared by a simple substitution reaction of fluorine with fluorobenzene.
C6H5F + F2 → C6H4F2 + HFThe 1,4-isomer and small amounts of the 1,3-isomer are also produced in the reaction as the fluorine group on the aromatic ring of fluorobenzene is ortho- and para- directing.
Applications
1,2-Difluorobenzene has been used as solvent for the electrochemical analysis of transition metal complexes. It is relatively chemically inert, weakly coordinating, and has a dielectric constant high enough to dissolve many electrolytes and metal complex salts. It is used as a weakly coordinating solvent for metal complexes, alternative to the relatively more strongly coordinating solvents acetonitrile, DMSO, and DMF.
1,2-Difluorobenzene can be acylated to 3',4'-difluoropropiophenone, which has interesting application in the synthesis of halogenated cathinone/PPA congeners.
